About our Human Tissue Assays
REPROCELL is the leading CRO offering a wide range of safety, efficacy, and ADME assays in human fresh tissues.
Our clinical network, growing since 2002, provides ethically approved access to biospecimens for use in our research laboratories in the US and UK, where we have worked on over 1,000 potential new drugs to evaluate their effects in humans. We offer organotypic assays across a wide range of tissue types from both healthy and diseased tissues, covering diseases such as IBD, psoriasis, psoriasis, atopic dermatitis, COPD, asthma, hypertension, and heart failure. We can also offer comparative studies across species to better understand the translation of animal data to humans.
Data from patient derived explants (PDE) can be used to improve clinical success, by testing potential new drugs in the closest possible model to patients and can also add enormous commercial value to assets by demonstrating proof of concept in living biospecimens. We have worked with over 150 Pharma and Biotech companies – see our publications page for examples.
Drug Discovery Assay Catalog
REPROCELL Biopta is home to the world's first and largest human fresh tissue assay catalog. In this digital platform, you can filter our assays by tissue type, endpoint, receptor, platform technology, drug type, and more.
Assay Types
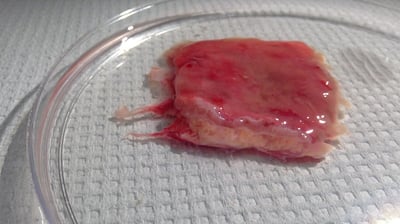
IBD
Inflammatory bowel diseases (IBD), such as ulcerative colitis and Crohn’s disease, have become a key focus for Pharma in recent years. Our scientists have extensive experience with efficacy, contractility, safety, ex vivo cultures, and ADME assays using intestinal tissue. We can access tissues from patients suffering from IBD, including Crohn’s disease and ulcerative colitis to investigate drug effects on intestinal permeability and inflammation.
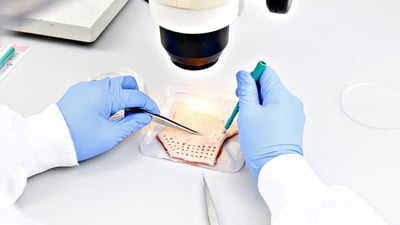
Skin Disease
Skin diseases represent a major health concern in the world today. Psoriasis affects more than 100 million people worldwide and is responsible for twice the disability burden caused by acute hepatitis C. We provide translational products and services to develop new therapies for serious skin diseases, including psoriasis, acne, and atopic dermatitis. Several inflammatory skin disease models are available to suit your research needs and budget, including patient biopsies, induced models, and 3D models.

ADME/DMPK
Accurately predicting the oral bioavailability of your test articles is key to clinical success. Our ADME studies use human tissues to estimate drug permeability, metabolism, or ion channel function ex vivo - giving you the most accurate prediction of oral bioavailability before clinical trial. With our Ussing chamber system, we can measure the effects of your test article on gastrointestinal permeability, gastrointestinal metabolism, and ion channel function.
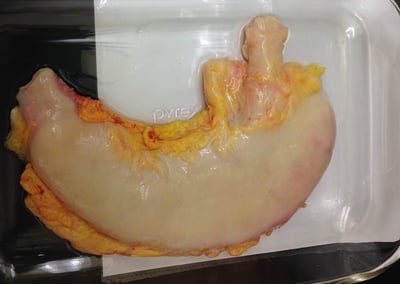
Gastrointestinal Motility
A change in gastrointestinal (GI) motility can result in a spectrum of symptoms, including heartburn, diarrhea, and gastroparesis. We can investigate the effect of your article on GI motility in a range of human tissues, including the stomach, intestines and lower esophageal sphincter. Determine the therapeutic potential of your compound, or investigate adverse clinical effects using our fresh ex vivo assays.
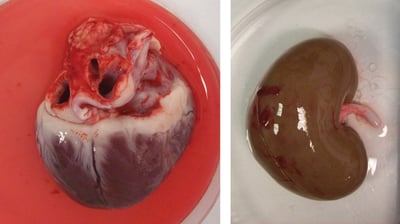
Species Comparisons
If you want to translate your data from animal models, we can directly compare human and animal data in our ex vivo models. Our species comparison assays provide an improved understanding of species differences in response to your test article and are commonly used to troubleshoot clinical adverse effects in respiratory, cardiovascular, genitourinary, and ADME/DMPK studies.
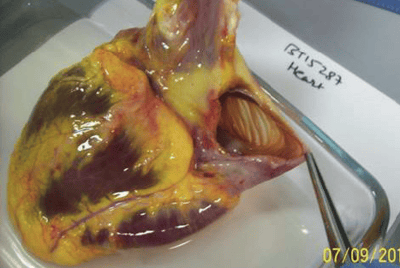
Cardiovascular
As a leading cause of death in the developed world, Cardiovascular disease (CVD) continues to be a major focus of drug development. However, even if you are not a CVD researcher, you may be interested in negating any off-target cardiovascular effects of your test articles. We offer assays in numerous cardiovascular tissues including cardiac muscle, arteries, and veins.
Respiratory
Respiratory diseases are some of the most common medical conditions in the world. Chronic obstructive pulmonary disease (COPD) accounts for 5% of global deaths annually, while asthma is the most common non-communicable disease in children. We offer an extensive range of assays in fresh human lung tissue from non-transplantable lungs or surgical residual tissues, including precision-cut lung slices (PCLS), pulmonary arteries, airway contractility, and lung parenchymal culture.
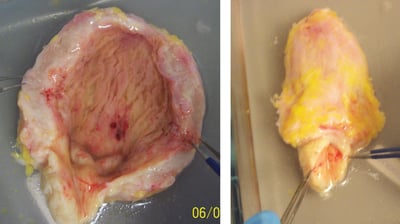
Genitourinary
Genitourinary disorders include congenital anomalies, infectious diseases, trauma, or conditions that involve the urinary structure or function. Our scientists have extensive experience assessing contractile and relaxation responses in these tissues, including the uterus, ureter, urethra, prostate, and bladder.
Neuronal
With the global aging population, neurodegenerative diseases are becoming increasingly important due to their huge cost burden on health care. Our organotypic neurite outgrowth model can be used to explore a wide range of experimental end-points in healthy or diseased human neurons. We also provide iPSC-derived human neurocytes and custom-differentiated neurons.




![Wire myography: the ultimate guide [protocol included]](https://www.reprocell.com/hs-fs/hubfs/REPROCELL-04.06.18_0163.jpg?width=756&height=425&name=REPROCELL-04.06.18_0163.jpg)